
Two of the largest pharmaceutical markets, the US and Japan, show distinct drug approval trends for Alzheimer’s disease (AD) compared to other key regions. Both countries have approved only the same two monoclonal antibodies (mAbs) alongside no small molecules. This suggests a selective and stricter regulatory approach, which converges on a narrower acceptance for AD drug approval, according to GlobalData, a leading intelligence and productivity platform.
AD is a brain disorder that slowly destroys memory and thinking skills. Eventually, AD patients lose the ability to perform simple tasks such as eating or walking. Age is the biggest risk factor for AD, and the current estimates suggest more than six million Americans suffer from the condition.
Within the eight major markets (8MM) (the US, France, Germany, Italy, Spain, the UK, Japan, and China), there are only six innovator drugs approved for AD.
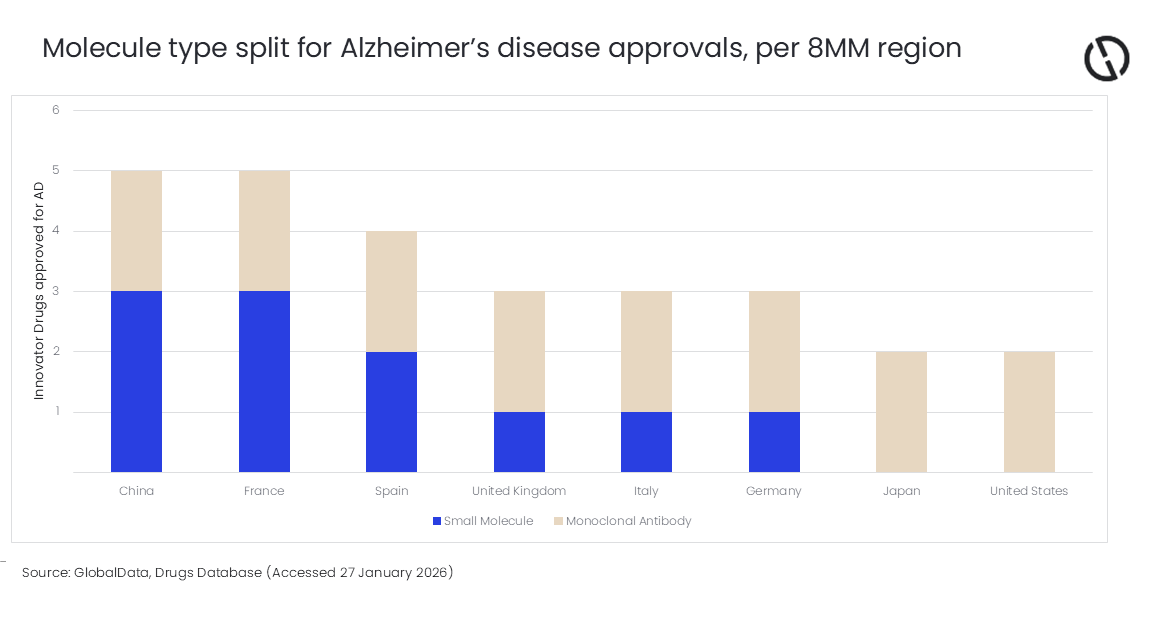
Jasper Morley, Pharma Analyst at GlobalData, comments: “All eight geographies have approved the same two mAbs, Eisai’s Leqembi (lecanemab) and Eli Lilly’s Kisunla (donanemab), suggesting a global acceptance of these products. Despite a larger pool of pipeline small molecules under development and available for approval, there is significant regional variety in the number of small molecules approved.”
China and France have both approved three small molecules for AD, while the remaining European countries have all approved at least one small molecule. Two of these small molecules, donepezil and memantine, are older therapies, and received approval in these regions in the late 1990s and early 2000s, with France later approving an orally dissolving version of donepezil in 2006. Alternatively, novel small molecules like sodium oligomannate received Chinese approval in 2019.
Morley continues: “The US and Japan present significantly different AD approval trends, having not approved any small molecules. Given these regions represent two of the world’s largest pharmaceutical markets, this narrower landscape is unlikely to reflect bandwidth or financial incapabilities. This absence of small molecule approvals in the US and Japan suggests a harder regulatory line for this molecule type for AD, in comparison to their market peers.
“Ultimately, this leaves patients with fewer therapeutic options available, which may be especially impactful in Japan, where the society is significantly aged. Nevertheless, the US and Japanese approval patterns align with a broader industry shift towards biologics over small molecules.”
The approved mAbs in AD, Leqembi and Kisunla, have showcased limited efficacy in practice, with the UK’s National Institute of Health and Care Excellence (NICE) refusing to recommend either drug for the National Health Service (NHS), citing high costs and limited benefits.
Morley concludes: “With 10 small molecules in Phase III, and soon to be seeking approval in Japan or the US, it is possible we could see several novel entries into these markets, should these products in development demonstrate the clinical benefit required for approval. Such approvals would mark a significant shift in the current treatment paradigm, broadening therapeutic options beyond mAbs for patients in these regions.”
See What’s Next in Tech With the Fast Forward Newsletter
Tweets From @varindiamag
Nothing to see here - yet
When they Tweet, their Tweets will show up here.





























